 
The lightest element was hydrogen (atomic number 1) and the heaviest element was uranium (atomic number 92). The first modern periodic table was created in 1869. Copper and chlorine are slightly unusual in that they both have two relatively common isotopes. All of the other elements listed in the table above have isotopes that are even more common (practically 100%). 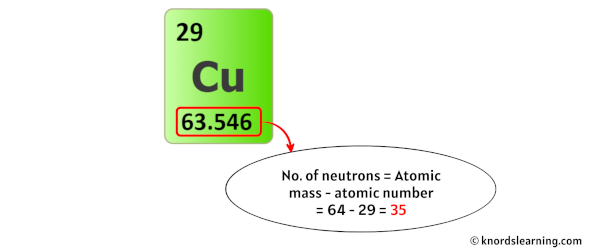
98.94% of the carbon atoms on Earth are 12C, 99.64% of the nitrogen atoms are 14N, 99.76% of the oxygen atoms are 16O, 75.76% of the chlorine atoms are 35Cl, 96.94% of the calcium atoms are 40Ca, and 91.75% of the iron atoms are 56Fe. 99.99% of the hydrogen atoms on Earth are 1H, so when we talk about the “hydrogen” atom, we usually mean 1H. It is easy to get lazy and ignore the fact that most types of atoms can exist as several different isotopes because one of those isotopes is usually far more common than the others. However, the actual mass of a mole of copper will depend on the composition of the specific sample. One mole of copper will therefore have a combination of both copper-63 atoms and copper-65 atoms in it, and the “molar” mass of copper is considered to be 63.546 g/mol. Copper-65 has a greater mass than copper-63 because it has two more neutrons in its nucleus. It has a mass number of 65 (29 protons and 36 neutrons) and an atomic mass of 64.9278 u. Most of the remaining 31% of copper atoms on Earth are copper-65 ( 65Cu). It has a mass number of 63 (29 protons and 34 neutrons) and an atomic mass of 62.9296 u. About 69% of the copper atoms on Earth are copper-63 (this percentage may vary in other solar systems and galaxies). Copper-63 ( 63Cu) is the most common isotope. 
Copper can exist as 29 different isotopes, and two of those isotopes are stable. There is not just one type of copper atom. Both protons and neutrons have masses approximately equal to 1 u (1 proton ≈ 1.0073 u, 1 neutron ≈ 1.0087 u, and 1 u ≈ 1.66 × 10 -27 kg), so the mass of a copper atom will be approximately 63 u, and the mass of 1 mole (6.022 × 10 23 atoms) of copper will have a mass of approximately 63 g. Because almost all of an atom’s mass is in its nucleus, we can estimate an atom’s mass just by counting its protons and neutrons. A neutrally charged copper atom will then have 29 electrons in orbit around its nucleus (29 − 29 = 0). This means that the copper atom has 29 protons and 34 neutrons in its nucleus (29 + 34 = 63). The atomic number is the number of protons in the nucleus, and the mass number is the number of protons and neutrons in the nucleus. This makes it easier to understand and predict how atoms will interact to form chemical bonds.Copper has an atomic number of 29 and a mass number of 63. The configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. This give us the (correct) configuration of:įor the Cu+ ion we remove one electron from 4s1 leaving us with:įor the Cu2+ ion we remove a total of two electrons (one from the 4s1 and one form the 3d10) leaving us with Therefore, one of the 4s2 electrons jumps to the 3d9. 
Half-filled and fully filled subshell have got extra stability. Therefore we have (still incorrect) 1s 22s 22p 63s 23p 63d 94s 2Ĭorrect Electron Configuration for Copper (Cu) Both of the configurations have the correct numbers of electrons in each orbital, it is just a matter of how the electronic configuration notation is written ( here is an explanation why). Note that when writing the electron configuration for an atom like Cu, the 3d is usually written before the 4s. Therefore the expected electron configuration for Copper will be 1s 22s 22p 63s 23p 64s 23d 9. After the 4s is full we put the remaining six electrons in the 3d orbital and end with 3d9. We now shift to the 4s orbital where we place the remaining two electrons. Since the 3s if now full we'll move to the 3p where we'll place the next six electrons. We'll put six in the 2p orbital and then put the next two electrons in the 3s. The p orbital can hold up to six electrons. The next six electrons will go in the 2p orbital. Since 1s can only hold two electrons the next 2 electrons for Copper go in the 2s orbital. In writing the electron configuration for Copper the first two electrons will go in the 1s orbital. Video: Cu, Cu +, and Cu 2+ Electron Configuration Notation 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
